To identify the effect of risk factors on diabetic patients. a study
was conducted among diabetic patients attending the outdoor
at the Sheikh Zaid Hospital, Lahore. Data was collected by interviewing
the patients using a structured questionnaire after the
approval of synopsis. SPSS 23.0 was used for data entry and analysis. A
sample of 100 respondents was selected by nonprobability
convenient sampling. The risk factors were analyzed in a gender study of
100. Tabular form was used to represent the
finding. Graphs shows the response of respondents. The Chi-Square test
has been used to assess the statistical significance of risk
factors for the diabetic patients. The check the normality of risk
factors and then apply Mann-Whitney test to check the effect of each
risk factor on diabetic patients w.r.t gender and marital status. The
result found that in sheikh Zaid hospital patients only physical
exercise, complications and environmental factors are affected in
diabetic patients.
Keywords: Diabetic patient; questionnaire; risk factors; chi square test; mann whitney test
Introduction
Diabetes mellitus
The word “diabetes” stems from a Greek term for passing through, a
reference to increased urination (polyuria), a common symptom of the
disease. “Mellitus” is the Latin word for honeyed, a reference to
glucose noted in the urine of diabetic patients. Diabetes mellitus is
sometimes referred to as sugar diabetes but usually is simply called
diabetes. Diabetes mellitus is a chronic disease caused by inherited or
acquired deficiency of insulin production or resistance to action of the
produced insulin. Diabetes occurs when the pancreas does not produce
enough insulin (a hormone that regulates blood sugar) or alternatively,
when the body cannot effectively use the insulin it produces. The
overall risk of dying among people with diabetes is at least double the
risk of their peers without diabetes (Setter et al., 2000). Insulin is
more of an anabolic hormone rather than catabolic. Insufficient amounts
of insulin or poor cellular response to insulin as well as defective
insulin leads to improper handling of glucose by body cells or
appropriate glucose storage in the liver and muscles. This ultimately
leads to persistently high levels of blood glucose, poor protein
synthesis, and other metabolic derangements. When there will be no
insulin production or insulin become resistant then glucose will not be
supply to the cells and remain as it is in the body. When it will not
utilize by the cells then glucose level elevates in the body and cause
hyperglycemic conditions in the body and the person is said to be
diabetic. Following may be the reason of increased level of glucose in
diabetic patients
- No production of insulin by pancreas
- Not enough insulin production that help in glucose supply to the cells
- Misfunctioning of insulin known as insulin resistance
The disease has been considered as one of the major health concerns
worldwide today. The increase in incidence of diabetes in developing
countries follows the trend of urbanization and lifestyle changes,
perhaps most importantly diet [1]. Diabetes Mellitus is the common
endocrine disease and affects nearly 10% of world population. At
present, 347 million people worldwide have diabetes. In 2004, an
estimated 3.4 million people died from consequences of fasting high
blood sugar. A similar number of deaths have been estimated for 2010.
More than 80% of diabetes deaths occur in low- and middle-income
countries . Many experts continued to advise strict carbohydrate
restriction, with the result that most people with diabetes adopted a
high fat, low carbohydrate diet. Diabetes mellitus (DM) could be a risk
factor for the development and progression of liver disease.
- Weight loss: Overly high blood sugar levels can
also cause rapid weight loss, say 10 to 20 pounds over two or three
months-but this is not a healthy weight loss. Because the insulin
hormone is not getting glucose into the cells, where it can be used as
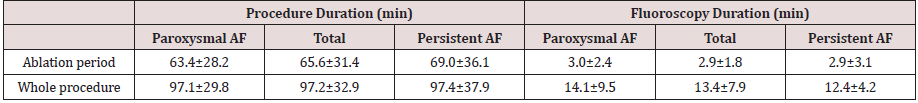
energy, the body thinks it's starving and starts breaking down protein
from the muscles as an alternate source of fuel.
- Hunger: Recessive pangs of hunger, another sign of
diabetes, can come from sharp peaks and lows in blood sugar levels. When
blood sugar levels plummet, the body thinks it has not been fed and
craves more of the glucose that cells need to function.
- Slow healing: Infections, cuts,
and bruises that do not heal quickly are another classic sign of
diabetes. This usually happens because the blood vessels are being
damaged by the excessive amounts of glucose traveling the veins and
arteries. This makes it hard for blood-needed to facilitate healing-to
reach different areas of the body.
- Increased urination, excessive thirst:
If you need to urinate frequently-particularly if you often must get up
at night to use the bathroom-it could be a symptom of diabetes. The
kidneys kick into high gear to get rid of all that extra glucose in the
blood, hence the urge to relieve yourself, sometimes several times
during the night. The excessive thirst means your body is trying to
replenish those lost fluids.
- Causes of diabetes:
The causes of diabetes are complex and only partly understood. This
disease is generally considered multifactorial, involving several
predisposing conditions and risk factors. In many cases genetics, habits
and environment may all contribute to a person’s diabetes. Weight and
body type, Family medical history, Lack of physical activity,
Carbohydrate intake, Chemical exposure, Smoking, Alcohol intake. This is
blamed largely on the rise of obesity and the global spread of
Western-style habits: physical inactivity along with a diet that is high
in calories, processed carbohydrates, and saturated fats and
insufficient in fiber rich whole foods. The aging of the population is
also a factor. However, other factors, such as environment may also be
contributing, because cases of autoimmune diabetes (type 1) are also
becoming more common [2-10]. Experts are urging people to help stem this
epidemic by getting regular exercise and controlling their diet and
weight. Humans are not the only species that can develop diabetes. This
disease also occurs in dogs, cats and other animals, as increasing
numbers of pet owners are discovering.
Diabetic complications
The direct and indirect effects on the human vascular tree are the
major source of morbidity and mortality in both type 1 and type 2
diabetes. Generally, the injurious effects of hyperglycemia are
separated into macrovascular complications (coronary artery disease,
peripheral arterial disease, and stroke) and microvascular complications
(diabetic nephropathy, neuropathy, and retinopathy). More than half of
all individuals with diabetes eventually develop neuropathy. Long-term
metabolic complications of diabetes mellitus include retinopathy,
nephropathy, peripheral neuropathy, amputations, and Charcot joints as
well as autonomic neuropathy causing gastrointestinal, genitourinary,
cardiovascular symptoms and sexual dysfunction. Diabetics are also at a
greater risk atherosclerotic, cardiovascular, peripheral arterial and
cerebrovascular disease. Hypertension and abnormalities of lipoprotein
metabolism also accompany uncontrolled diabetes mellitus. These
cardiovascular disorders are the leading cause of death in people with
diabetes. Diabetes is the chief cause of end-stage renal disease, which
requires treatment with dialysis or a kidney transplant. These include
diabetic retinopathy, glaucoma and cataracts. Diabetes is a leading
cause of visual impairment and blindness. This includes peripheral
neuropathy, which often causes pain or numbness in the limbs, and
autonomic neuropathy, which can impede digestion (gastroparesis) and
contribute to sexual dysfunction and incontinence. Neuropathy may also
impair hearing and other senses. Many studies have linked diabetes to
increased risk of memory loss, dementia, Alzheimer’s disease and other
cognitive deficits. Recently some researchers have suggested that
Alzheimer’s disease might be “type 3 diabetes,” involving insulin
resistance in the brain. Foot conditions and skin disorders, such as
ulcers, make diabetes the leading cause of nontraumatic foot and leg
amputations. People with diabetes are also prone to infections including
periodontal disease, thrush, urinary tract infections and yeast
infections [11-16]. Diabetes increases the risk of malignant tumors in
the colon, pancreas, liver and several other organs. Conditions ranging
from gout to osteoporosis to restless legs syndrome to myofascial pain
syndrome are more common in diabetic patients than nondiabetics.
Diabetes increases the risk of preeclampsia, miscarriage, stillbirth and
birth defects. Many but not all the studies exploring connections
between diabetes and mental illness have found increased rates of
depression, anxiety and other psychological disorders in diabetic
patients. In addition to chronic hyperglycemia, diabetic patients can
experience acute episodes of hyperglycemia as well as hypoglycemia (low
glucose).
Gestational diabetes
Gestational diabetes mellitus (GDM) is defined as any degree of
glucose intolerance with onset or first recognition during pregnancy.
The definition applies whether insulin or only diet modification is used
for treatment and whether the condition persists after pregnancy.
Approximately 7% of all pregnancies are complicated by GDM, resulting in
more than 200,000 cases annually.
Type 1 diabetes
In type 1 diabetes, hyperglycemia occurs because of a complex disease
process where genetic and environmental factors lead to an autoimmune
response that remains to be fully elucidated. During this process, the
pancreatic B-cells within the islets of Langerhans are destroyed,
resulting in individuals with this condition relying essentially on
exogenous insulin administration for survival, although a subgroup has
significant residual C- peptide production. Type 1 diabetes is a disease
in which the pancreas does not produce any insulin. Insulin is a hormone
that helps your body to control the level of glucose (sugar) in your
blood. Without insulin, glucose builds up in your blood instead of being
used for energy. Your body produces glucose and gets glucose from foods
like bread, potatoes, rice, pasta, milk, and fruit. An autoimmune
disease in which the immune system mistakenly destroys the
insulin-making beta cells of the pancreas. It typically develops more
quickly than other forms of diabetes. It is usually diagnosed in
children and adolescents, and sometimes in young adults. To survive,
patients must administer insulin medication regularly. This form of
diabetes previously encompassed by the terms insulin–dependent diabetes,
Type 1 diabetes, or juvenile– onset diabetes, results from autoimmune
mediated destruction of the beta cells of the pancreas. The rate of
destruction is quite variable, being rapid in some individuals and slow
in others. The rapidly progressive form is commonly observed in
children, but also may occur in adults. The slowly progressive form
generally occurs in adults and is sometimes referred to as latent
autoimmune diabetes in adults (LADA) [17-26]. Markers of immune
destruction, including islet cell autoantibodies, and/or autoantibodies
to insulin, and autoantibodies to glutamic acid decarboxylase (GAD) are
present in 85–90 % of individuals with Type 1 diabetes mellitus when
fasting diabetic hyper glycaemia is initially detected.
Type 2 diabetes
Type 2 diabetes is the result of failure to produce sufficient
insulin and insulin resistance. Elevated blood glucose levels are
managed with reduced food intake, increased physical activity, and
eventually oral medications or insulin. Type 2 diabetes is believed to
affect more than 15 million adult Americans, 50% of whom are
undiagnosed. It is typically diagnosed during adulthood. However, with
the increasing incidence of childhood obesity and concurrent insulin
resistance, the number of children diagnosed with type 2 diabetes has
also increased worldwide Type 2 diabetes is Caused by insulin resistance
in the liver and skeletal muscle, increased glucose production in the
liver, over production of free fatty acids by fat cells and relative
insulin deficiency. Insulin secretion can be decreases with gradual
failure of beta cells.
Contributing factors of type 2 diabetes: Obesity,
Age (onset of puberty is associated with increased insulin resistance)
Lack of physical activity, Genetic predisposition, Racial/ethnic
background (African American, Native American, Hispanic and
Asian/Pacific Islander), Conditions associated with insulin resistance,
(e.g., polycystic ovary syndrome).
Causes of type 2 diabetes: Obesity,
Excess glucorticoid, Excess growth hormone, Gestational diabetes,
Polycystic ovary disease, Lipodystrophy, Mutation of insulin receptor,
Hemochromatosis, Blurry vision, Tingling or numbness. The most
significant contributors to or causes of type 2 diabetes are diet and
exercise. Obesity is a major risk-factor for diabetes.
Blurry vision: Having distorted vision and seeing
floaters or occasional flashes of light are a direct result of high
blood sugar levels. "Blurry vision is a refraction problem. Diabetes
mellitus is group of metabolic disorders characterized by hyper
glycemia, glycosuria and hyperlipemia”. In 2000 almost 177 million
inhabitants of the world were affected by diabetes and in future (2025)
predictable range of the people which are going to effect by the
diabetes is 300 million. Type 2 diabetes is the type of diabetes in
which insulin is produced but cells don’t take insulin for glucose
uptake. Inactive sittings, fatness is the main cause of type 2 diabetes.
Diabetes is a global problem, and its occurrence is continuously
increasing in the world. Pakistan is at 7th rank in list of countries
and it expected to have on 4th rank in future. Therefore, for research
purpose diabetes is selected because the ratio of this disease is
continuously increasing. Serum samples were collected from Sheik Zayed
hospital Lahore because this hospital was nearer to Punjab University
Lahore and have a separate diabetes department.
Methodology:
Study design: It was a cross-sectional study.
Setting: The Study was conducted at Diabetes Centre, Sheikh Zaid Hospital Lahore.
Selection of hospital: Shaikh Zayed
Hospital is a tertiary care hospital located in Lahore, Punjab,
Pakistan. It is attached with Shaikh Khalifa Bin Zayed Al-Nahyan Medical
and Dental College as a teaching hospital and is part of Shaikh Zayed
Medical Complex Lahore. And hospital is under Government of Pakistan.
Their management will be very fine as compared other government
hospitals. People believes that hospital is better than other so mostly
people are coming in this hospital for their treatments so that why I
use this hospital.
Target Population: All the patient came to the outpatient diabetes department of Sheikh Zaid Hospital Lahore. Who have Type 2 diabetes?
Duration of study: The duration of study was two months (02-05-2018 to 02-07-2018) after the approval of synopsis.
Sample Selection: Sample selection
is one of the most vital steps for conducting a research. As the
conclusion of the study is based on sample and all the inference are
consequently referred to whole the population it should be a good
representative to the target population.
Sampling technique: Non-probability convenient sampling technique was used for collection of data.
Sample size: 1000 cases were used in this study.
Data collection procedure: The
success of the survey depends upon accuracy of the data collection. The
correction of the accurate data depends upon the correct choice of
survey method. After questionnaire, the next step was data collection.
Face to face method was used for the collection of data keeping in mind
the difficulty of locating the respondent after giving them the
questionnaire. So, it was the best way to give the questionnaire to the
respondent and be there for a while until the respondent fill and give
it back. Respondent asks the purpose of the survey, meaning of the
questions which they do not completely understand. Data were collected
by suing a Performa/Questionnaire. The first part of the Performa
contained information’s about the demographic characteristic of the
patients while the second part contained information regarding risk
factors of the disease. The collection of the accurate data depends upon
the careful construction of a tool of data collection. There are some
difficulties in field experience [27-34]. The respondent’s behavior was
good, but some respondents refused to fill up the questionnaire. After
explaining the objective of the study, they agreed to cooperate. Though
at some places of the behavior of the respondents were not encouraging
but it was a great experience overall.
- Inclusion criteria: The patient came to the outpatient diabetes department agreed to provide information.
- Exclusion criteria: Patients who are not agreeing to provide information.
Data Analysis:
Software package: Data were entered and analyzed by using SPSS (Statistical Package for Social Science) version 23.
Statistical Technique
- Descriptive Analysis: For descriptive of variables
frequency were shown in tables. Charts and graphs were given for
percentages in qualitative variables.
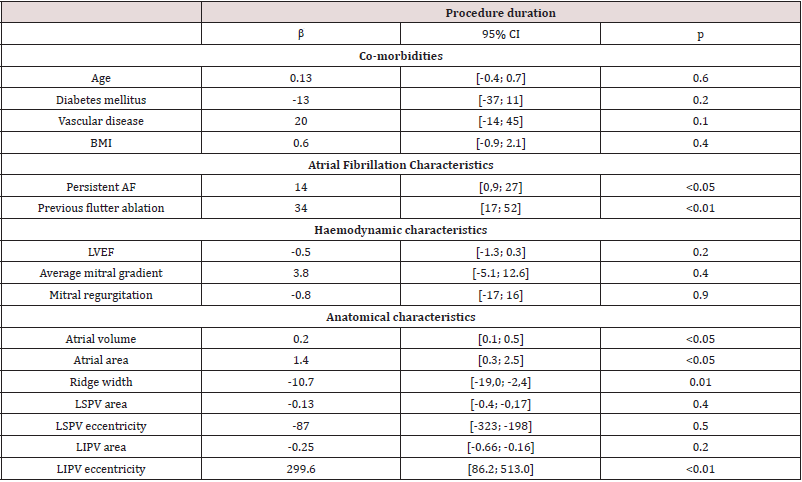
- Analytical Analysis: To find the risk variable of diabetes gender wise the current section is divided in the two main components.
- Bivariate Analysis
- Logistic Regression
Results
Figure 1: Shows the perecentage variation among the diabetic patients with various factors from figure 4.1.1 to 4.2.41.
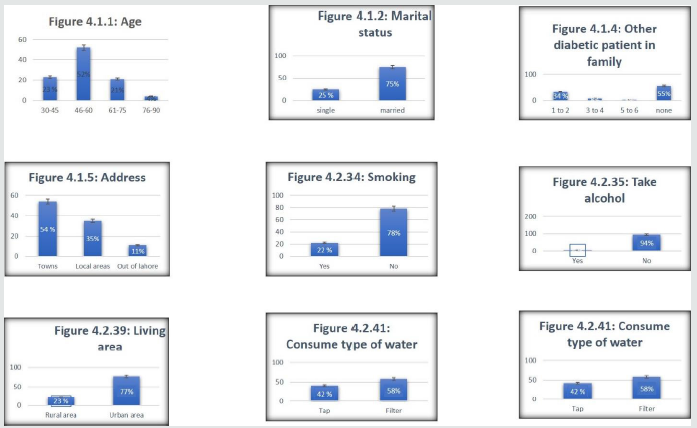
This study consists of 1000 subjects in which both male and female
are included. There are 53 variables age, other diabetic patients in
family, family members, address, marital status, gender, regarding
follow doctor, type of meal, skip meal, gain weight, vision problem,
wound problem, sugar fluctuation, social life, smoking, alcohol, alcohol
frequently use, sanitary area, regularly use of medicine, fact of
necessary exercise, fact of routine walk, daily walk, exercise, kind of
exercise, time of exercise, day spend in exercise, walking time, Meals,
hoteling, frequently of hoteling, use of fruits, use of milk, take care
of yourself, loss weight, kidney problem, skin problem, regularly check
sugar, sugar check time in a day, sugar record, sugar level, routine
work, hobbies, effect of diabetes, industry area, industry type, living
area, type of water, kind of medicine, use of vitamins, check-up,
discuss problem with doctor, satisfaction from treatment. Figure 1 shows
that out of 1000 respondents, 23(23.0%) persons have 30-45 age,
52(52.0%) persons have 46-60, 21(21.0%) persons have 61-75 and 4(4.0%)
persons have 76-90. Among 23 persons who have the 30-45 age, the count
(percentages) for male and female were 7(30.4%) and 16(69.6%)
respectively and among 52 persons who have the 46-60 age, the count
(percentages) for male and female were 16(30.8%) and 36(69.2%)
respectively and among 21 persons who have 76-90 age , the count
(percentages) for male and female were 4(100.0%) and 0(0.0%)
respectively . Figure 1 shows that out of 1000 respondents, 25 (25.0%)
persons have single while 75(75.0%) persons have married. Among 25
persons who are single , the count (percentages) for male and females
were 11(44.0) and 14(56.0%) respectively and among 75 persons who are
married, the count (percentages) for male and females were 28(37.3%) and
47(62.7%) respectively. Figure 1 shows that of out of 1000 respondents,
37(37.0%) persons have 1-5 family members, 47(47.0%) persons have 6-10
family members, 12(12.0%) have 11-15 family members and 4(4.0% have
16-20 family members. Among 37 persons have 1-5 family members, the
count (percentages) for male and females were 18(49.6%) and 19(51.4%)
respectively and among 47 persons have 6-10 family members, the count
(percentages) for male and females were 19(40.4%) and 28 (59.6%)
respectively and among 12 persons have 11-15 family members, the count
(percentages) for male and females were 1(8.3%) and 11(91.7%)
respectively and among 4 persons have 16-20 family members, the count
(percentages) for male and females were 1(25.0%) and 3(75.0%). Figure 1
shows that of out of 1000 respondents, 34(34.0%) persons have 1-2
diabetic patient in family members, 8(8.0%) persons have 3-4 diabetic
patient in family members, 3(3.0%) have 5-6 diabetic patient in family
members and 55(55.0%) have no diabetic patient in family members. Among
34 persons have 1-2 diabetic patient in family members, the count
(percentages) for male and females were 12(35.3%) and 22(64.7%)
respectively and among 8 persons have 3-4 diabetic patient in family
members, the count (percentages) for male and females were 0(0.0%) and
8(100.0%) respectively and among 55 persons have no diabetic patient in
family members, the count (percentages) for male and females were
27(49.1%) and 28(50.9%) respectively [35-46]. Figure 1 shows that of out
of 1000 respondents, 54(53.0%) persons address of towns, 3535.0%)
persons have address of local areas and 11(11.0%) persons address out of
Lahore. Among 54 persons address of towns, the count (percentages) for
male and females were 20(37.0%) and 34(63.0%) respectively and among 35
persons address of local areas, the count (percentages) for male and
females were 16(45.7%) and 19(54.3%) respectively and among 11 persons
address of out of Lahore, the count (percentages) for male and females
were 3(27.3%) and 8(72.7%) respectively Figure 1 shows that of out of
1000 respondents, 22(22.0%) persons that are doing smoking and 78(78.0%)
persons that are not doing smoking. Among 22 that are doing smoking,
the count (percentages) for male and female were 20(90.0%) and 2(9.1%)
respectively and among 78 persons that are not doing smoking, the count
(percentages) for males and females were 19(24.4%) and 59(75.6%)
respectively Figure 1 shows that of out of 1000 respondents, 6(6.0%)
persons that are taking alcohol and 94(94.0%) persons that are not
taking alcohol. Among 6 that are taking alcohol, the count (percentages)
for male and female were 6(100.0%) and 0(0.0%) respectively and among
94 persons that are not taking alcohol, the count (percentages) for
males and females were 33(35.1%) and 61(64.5%) respectively Figure 1
shows that of out of 1000 respondents, 23(23.0%) persons that are living
in rural area and 77(77.0%) persons that are living in urban area
[47-53]. Among 23 that are living in rural area, the count (percentages)
for male and female were 6(26.1%) and 17(73.9%) respectively and among
77 persons that are living in urban area, the count (percentages) for
males and females were 33(42.9%) and 44(57.1%) respectively. Figure 2
shows that of out of 1000 respondents, 27(27.0%) persons that their area
sanitary system is very good and 45(45.0%) persons that their area
sanitary system is good and 17(17.0%) persons that there are a sanitary
system is bad and 11(11.0%) persons that their area sanitary system is
very bad. Among persons that their area sanitary system is very good,
the count (percentages) for male and female were 4(14.8%) and 23(85.2%)
respectively and among 45 persons that their area sanitary system is
good, the count (percentages) for males and females were 27(60.0%) and
18(40.0%) respectively and among persons that there are a sanitary
system is bad, the count (percentages) for male and female were 7(41.2%)
and 10(58.8%) and 11 persons that their area sanitary system is very
bad, the count (percentages) for male and female were 1(9.1) and
10(90.9%) respectively Figure 1 shows that of out of 1000 respondents,
42(42.0%) persons that consume tap water and 58(58.0%) persons that are
consume filter water. Among 42 that are consume tap water, the count
(percentages) for male and female were 15(35.7%) and 27(64.3%)
respectively and among 58 persons that are consume filter water, the
count (percentages) for males and females were 24(40.0%) and 36(60.0%)
respectively. Figure 2 shows that of out of 1000 respondents, 25(25.0%)
persons that are living in industrial area and 75(75.0%) persons that
are not living in industrial area. Among 26 that are living in
industrial area, the count (percentages) for male and female were
10(40.0%) and 15(60.0%) respectively and among 75 persons that are not
living in industrial area, the count (percentages) for males and females
were 29(38.7%) and 46(61.3%) respectively Figure 2 shows that of out of
1000 respondents, 80(80.0%) persons think that exercise is necessary
for diabetic patients, 17(17.0%) persons thought that exercise is not
necessary for diabetic patients and 3(3.0%) persons have no idea that
exercise is suitable or not for diabetic patients. Among 80 persons
think that the exercise is necessary for diabetic patients, the count
(percentages) for male and females were 31(38.8%) and 49(61.3%)
respectively and among 17 persons think that exercise is not necessary
for diabetic patients, the count (percentages) for male and females were
6(35.3%) and 11(64.7%) respectively and among 3 persons don’t know that
exercise is necessary for diabetic patients, the count (percentages)
for male and females were 2(66.7%) and 1(33.3%) respectively. Figure 2
shows that of out of 1000 respondents, 88(88.0%) persons think that
routine Walk is helpful for diabetic patients, 9(9.0%) persons think
that walk is not helpful for diabetic patients and 3(3.0%) persons have
no idea that walk is helpful or not. Among 88 persons think that the
routine walk is helpful for diabetic patients, the count (percentages)
for male and females were 32(36.4%) and 56(63.6%) respectively and among
9 persons think that routine is not helpful for diabetic patients, the
count (percentages) for male and females were 5(55.6%) and 4(44.4%)
respectively and among 3 persons don’t know that routine walk is helpful
or not for diabetic patients, the count (percentages) for male and
females were 2(66.7%) and 1(33.3%) respectively.
Figure 2: this shows percentage variation in pi charts form from 4.2.1 to 4.2.11.
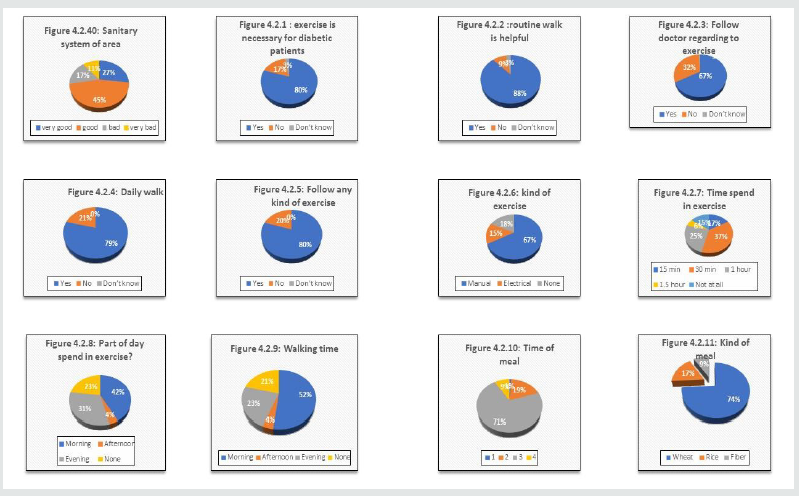
Figure 2 shows that of out of 1000 respondents, 67(67.0%) persons
follow doctor regarding to exercise, 32(32.0%) persons do not follow
doctor regarding to exercise, and 1(1.0%) persons don’t know about
follow doctor regarding to exercise. Among 67 persons follow doctor
regarding to exercise, the count (percentages) for male and females were
21(31.3%) and 46(68.7%) respectively and among 32 persons don’t follow
doctor regarding to exercise, the count (percentages) for male and
females were 18(56.3%) and 14(43.8%) respectively and among 1 persons
don’t know about follow doctor regarding to exercise, the count
(percentages) for male and females were 0(0.0%) and 1(100.0%)
respectively Figure 2 shows that of out of 1000 respondents, 79(79.0%)
persons go for daily walk, 21(21.0%) persons do not go for daily walk.
Among 79 persons go for daily walk, the count (percentages) for male and
females were 34(43.0%) and 45(57.0%) respectively and among 21 persons
do not go for daily walk, the count (percentages) for male and females
were 5(23.8%) and 16(76.2%) respectively. Figure 2 shows that of out of
1000 respondents, 80(80.0%) persons follow any kind of exercise,
20(20.0%) persons do not follow any kind of exercise. Among 80 persons
follow any kind of exercise, the count (percentages) for male and
females were 31(38.8%) and 49(61.3%) respectively and among 20 persons
do not follow any kind of exercise, the count (percentages) for male and
females were 8(40.0%) and 12(60.0%) respectively Figure 2 shows that of
out of 1000 respondents, 67(67.0%) persons that follow manual exercise ,
15(15.0%) persons that follow electrical exercise and 18(18.0) persons
that don’t follow any manual or electrical exercise. Among 67 persons
that follow manual exercise, the count (percentages) for male and
females were 25(37.3%) and 42(62.7%) respectively and among 15 persons
that follow electrical exercise, the count (percentages) for male and
females were 6(40.0%) and 9(60.0%) respectively and among 18 persons
don’t follow any manual or electrical exercise, the count (percentages)
for male and females were 8(44.4%) and 10(55.6%) respectively Figure 2
shows that of out of 1000 respondents, 17(17.0%) persons that spend time
in exercise 15 min, 37(37.0%) persons that spend time in exercise 30
min, 25(25.0) persons that spend time in exercise 1 hour , 6(6.0)
persons that spend time in exercise 1.5 hour and 15(15.0) persons that
spend no time on exercise. Among 17 persons that spend time in exercise
15 min, the count (percentages) for male and females were 3(17.6%) and
14(82.4%) respectively and among 37 persons that spend time in exercise
30 min, the count (percentages) for male and females were 19(51.4%) and
18(48.6%) respectively and among 25 persons that spend time in exercise 1
hour, the count (percentages) for male and females were 9(36.0%) and
16(64.0%) respectively and among 6 persons that spend time in exercise
1.5 hour, the count (percentages) for male and females were 1(16.7%) and
5(83.3%) respectively and among 15 persons that spend no time in
exercise, the count (percentages) for male and females were 7(46.7%) and
8(53.3%) respectively
Figure 2 shows that of out of 1000 respondents, 42(42.0%) persons
that spend morning in exercise, 4(4.0%) persons that spend afternoon in
exercise, 31(31.0) persons that spend evening in exercise and 23(23.0%)
persons spend no part of day in exercise. Among 42 persons that spend
morning in exercise, the count (percentages) for male and females were
15(36.7%) and 27(64.3%) respectively and among 4 persons that spend
afternoon in exercise, the count (percentages) for male and females were
2(50.0%) and 2(50.0%) respectively and among 31 persons that spend
evening in exercise, the count (percentages) for male and females were
11(35.5%) and 20(64.5%) respectively and among 23 persons spend no part
of day in exercise, the count (percentages) for male and females were
11(47.8%) and 12(52.2%) respectively. Figure 2 shows that of out of 1000
respondents, 52(52.0%) persons that spend morning for walk, 4(4.0%)
persons that spend afternoon for walk, 23(23.0%) persons that spend
evening for walk and 21(21.0%) persons spend no time for walk. Among 52
persons that spend morning for walk, the count (percentages) for male
and females were 24(46.2%) and 28(53.8%) respectively and among 4
persons that spend afternoon for walk, the count (percentages) for male
and females were 1(25.0%) and 3(75.0%) respectively and among 23 persons
that spend evening for walk, the count (percentages) for male and
females were 8(39.1%) and 14(60.9%) respectively and among 23 persons
spend no time for walk, the count (percentages) for male and females
were 5(23.8%) and 16(76.2%) respectively. Figure 2 shows that of out of
1000 respondents, 1(1.0%) persons that 1 time take meal in day,
19(19.0%) persons that 2 times take meal in a day, 71(71.0%) persons
that 3 times take meal in a day, and 9(9.0%) persons that 4 times take
meal in a day. Among 1 persons that 1 time take meal in a day, the count
(percentages) for male and females were 0(0.0%) and 1(100.0%)
respectively and among 19 persons that 2 times take meal in a day, the
count (percentages) for male and females were 8(42.1%) and 11(57.9%)
respectively and among 71 persons that 3 times take meal in a day, the
count (percentages) for male and females were 25(35.2%) and 46(64.8%)
respectively and among 9 persons that 4 times take meal in a day, the
count (percentages) for male and females were 6(39.0%) and 3(33.3%)
respectively. Figure 2 shows that of out of 1000 respondents, 74(74.0%)
persons that use wheat in meal, 17(17.0%) persons that use rice in meal
and 9(9.0%) persons that use fiber in meal. Among 74 persons that use
wheat in meal, the count (percentages) for male and females were
33(44.6%) and 41(55.4%) respectively and among 17 persons that use rice
in meal, the count (percentages) for male and females were 2(11.8%) and
15(88.2%) respectively and among 9 persons that use fiber in meal, the
count (percentages) for male and females were 4(44.4%) and 5(55.6%)
respectively. Figure 3 shows that of out of 1000 respondents, 37(37.0%)
persons that go out for meal, 63(63.0%) persons that do not go out for
meal. Among 37 persons that go out for meal, the count (percentages) for
male and females were 15(40.5%) and 22(59.5%) respectively and among 63
persons that not go for meal, the count (percentages) for male and
females were 24(38.%) and 39(61.9%) respectively .
Figure 3 shows that of out of 1000 respondents, 64(64.0%) persons
that never go out for meal, 23(23.0%) persons that sometimes go out for
meal, 8(8.0%) persons that normally go out for meal, and 5(5.0%) persons
that have frequently go out for meal. Among 64 persons that never go
out for meal, the count (percentages) for male and females were
25(39.1%) and 39(60.9%) respectively and among 23 persons that sometimes
go out for meal, the count (percentages) for male and females were
7(30.4%) and 16(69.6%) respectively and among 8 persons that normally go
out for meal, the count (percentages) for males and females were
5(62.5%) and 3(37.5%) respectively and among 5 persons that frequently
go out for meal, the count (percentages) for male and female were
2(40.0%) and 3(60.0%). Figure 3 shows that of out of 1000 respondents,
41(41.0%) persons that regularly use of fruit, 20(20.0%) persons that
are not use of fruit, 39(39.0%) persons that sometimes use the fruits.
Among 41 persons that regularly use of fruit, the count (percentages)
for male and females were 16(39.0%) and 25(61.0%) respectively and among
20 persons that are not use of fruit, the count (percentages) for male
and females were 7(35.0%) and 13(65.0%) respectively and among 39
persons that sometime use of fruit, the count (percentages) for males
and females were 16(41.0%) and 23(59.0%) respectively Figure 3 shows
that of out of 1000 respondents, 48(48.0%) persons that regularly use of
milk, 18(18.0%) persons that are not use of milk, 34(34.0%) persons
that sometimes use the milk. Among 48 persons that regularly use of
milk, the count (percentages) for male and females were 21(43.8%) and
27(56.3%) respectively and among 18 persons that are not use of milk,
the count (percentages) for males and females were 4(22.2%) and
14(77.8%) respectively and among 34 persons that sometime use of milk,
the count (percentages) for males and females were 14(41.2%) and
20(58.8%) respectively Figure 3 shows that of out of 1000 respondents,
37(37.0%) persons that skip their meal, 34(34.0%) persons that are not
skip their meal, 29(29.0%) persons that response is don’t know means
that persons have not in mind that they skip meal or not in routine.
Among 37 persons that skip their meal, the count (percentages) for male
and females were 7(18.9%) and 30(81.1%) respectively and among 34
persons that are not skip their meal, the count (percentages) for males
and females were 19(55.9%) and 15(44.1%) respectively and among 29
persons that have not in mind that they skip meal or not in routine, the
count (percentages) for males and females were 13(44.8%) and 16(55.2%)
respectively.
Figure 3: This also shows the percentage variation in pi chart form from 4.2.12. to 4.2.40.
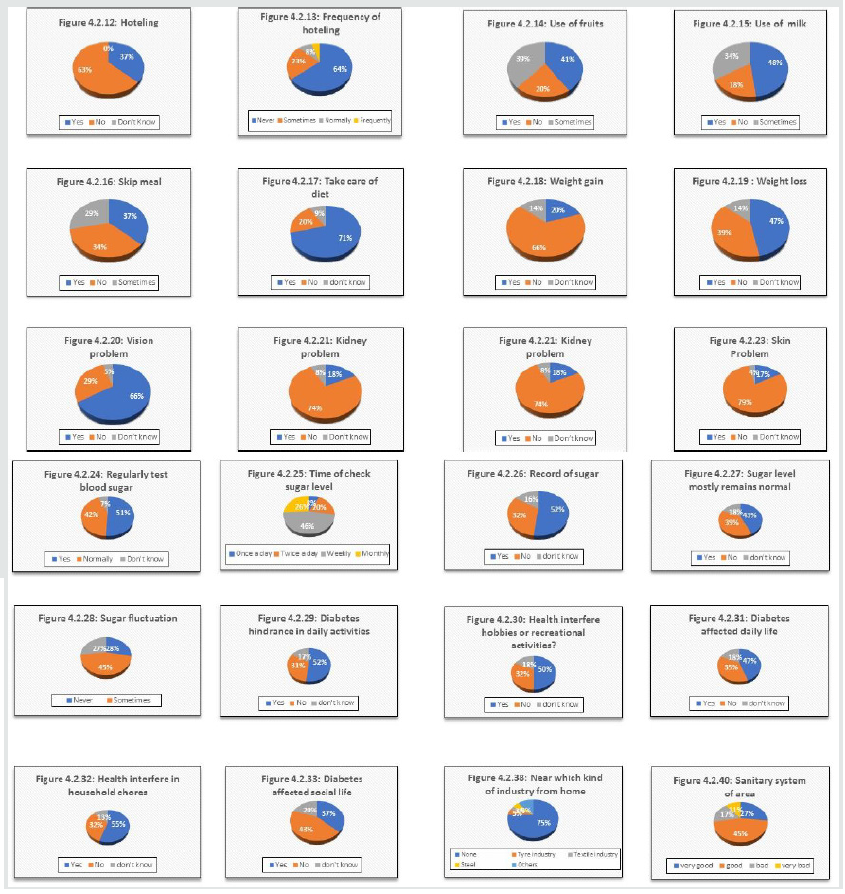
Figure 3 shows that of out of 1000 respondents, 71(71.0%) persons
that take of their diet, 20(20.0%) persons that are not take of their
diet, 9(9.0%) persons that response is don’t know means that persons
have not in mind that they take of diet or not. Among 71 persons that
skip their meal, the count (percentages) for male and females were
25(35.2%) and 46(64.8%) respectively and among 20 persons that are not
take of their diet, the count (percentages) for males and females were
11(55.0%) and 9(45.0%) respectively and among 9 persons that have not in
mind that they take of their diet or not, the count (percentages) for
males and females were 3(33.3%) and 6(66.7%) respectively. Figure 3
shows that of out of 1000 respondents, 20(20.0%) persons that weight
gain, 66(66.0%) persons that not weight gain, 14(14.0%) persons that
response is don’t know means that persons have not know that about their
weight that gain or not. Among 20 persons that gain weight, the count
(percentages) for male and females were 2(10.0%) and 18(90.0%)
respectively and among 66 persons that are not gain weight, the count
(percentages) for males and females were 31(47.0%) and 35(53.0%)
respectively and among 14 persons that don’t know that weight gain or
not, the count (percentages) for males and females were 6(42.9%) and
8(57.1%) respectively. Figure 3 shows that of out of 1000 respondents,
47(47.0%) persons that weight loss, 39(39.0%) persons that not weight
loss, 14(14.0%) persons that response is don’t know means that persons
don’t know that about their weight that loss or not. Among 47 persons
that loss weight, the count (percentages) for male and females were
18(38.3%) and 29(61.7%) respectively and among 39 persons that are not
lose weight, the count (percentages) for males and females were
15(38.5%) and 24(61.5%) respectively and among 14 persons that don’t
know that weight gain or not, the count (percentages) for males and
females were 6(42.9%) and 8(57.1%) respectively Figure 3 shows that of
out of 1000 respondents, 66(66.0%) persons that have vision problem,
29(29.0%) persons that have no vision problem and 5(5.0%) persons that
response is don’t know means that persons don’t know that about their
vision problem. Among 66 persons that have vision problem, the count
(percentages) for male and females were 18(27.3%) and 48(72.2%)
respectively and among 29 persons that have no vision problem, the count
(percentages) for males and females were 17(58.6%) and 12(41.4%)
respectively and among 5 persons that don’t know about their vision
problem, the count (percentages) for males and females were 4(80.0%) and
1(20.0%) respectively. Figure 3 shows that of out of 1000 respondents,
18(18.0%) persons that have kidney problem, 74(74.0%) persons that have
no kidney problem and 8(8.0%) persons that response is don’t know means
that persons don’t know that about their kidney problem. Among 18
persons that have kidney problem, the count (percentages) for male and
females were 4(22.2%) and 14(77.8%) respectively and among 74 persons
that have no kidney problem, the count (percentages) for males and
females were 30(40.5%) and 44(59.5%) respectively and among 8 persons
that don’t know about their kidney problem, the count (percentages) for
males and females were 5(62.5%) and 3(37.5%) respectively. Figure 3
shows that of out of 1000 respondents, 36(36.0%) persons that have wound
healing problem, 56(56.0%) persons that have no wound healing problem
and (8.0%) persons that response is don’t know means that persons don’t
know that about their wound healing problem. Among 36 persons that have
wound healing problem, the count (percentages) for male and females were
5(13.9%) and 31(86.1%) respectively and among 56 persons that have no
wound healing problem, the count (percentages) for males and females
were 29(51.8%) and 27(48.2%) respectively and among 8 persons that don’t
know about their wound healing, the count (percentages) for males and
females were 5(62.5%) and 3(37.5%) respectively.
Figure 3 shows that of out of 1000 respondents, 17(17.0%) persons
that have skin problem, 79(79.0%) persons that have no skin problem and
4(4.0%) persons that response is don’t know means that persons don’t
know that about their skin problem. Among 17 persons that have skin
problem, the count (percentages) for male and females were 5(29.4%) and
12(70.6%) respectively and among 79 persons that have no skin problem,
the count (percentages) for males and females were 32(40.5%) and
47(59.5%) respectively and among 4 persons that don’t know about their
skin problem, the count (percentages) for males and females were
2(50.0%) and 2(20.0%) respectively Figure 3 shows that of out of 1000
respondents, 51(51.0%) persons check their sugar level regularly,
42(42.0%) persons that are not check their sugar level regularly and
7(7.0%) persons that response is don’t know means that persons don’t
know that check their sugar level regularly. Among 51 persons that check
their sugar level regularly, the count (percentages) for male and
females were 15(29.4%) and 36(70.6%) respectively and among 42 persons
that are not check their sugar level regularly, the count (percentages)
for males and females were 22(52.4%) and 20(47.6%) respectively and
among 7 persons that don’t know about check their sugar level regularly,
the count (percentages) for males and females were 2(528.6%) and
5(71.4%) respectively Figure 3 shows that of out of 1000 respondents,
8(8.0%) persons check their sugar level once a day, 20(20.0%) persons
that check their sugar level twice a day, 46(46.0%) persons that check
their sugar level weekly and 8(8.0%) that check their sugar level
monthly. Among 8 persons that check their sugar level once a day, the
count (percentages) for male and female were 4(50.0%) and 4(50.0%)
respectively and among 20 persons that check their sugar level twice a
day, the count (percentages) for male and female were 7(35.0%) and
13(65.0%) respectively and among 46 persons that check their sugar level
weekly, the count (percentages) for male and female were 20(43.5%) and
26(56.5%) respectively and among 8 person that check their sugar level
monthly, the count(percentages) for male and female were 8(30.8%) and
18(69.2%) respectively. Figure 3 shows that of out of 1000 respondents,
52(52.0%) persons record their sugar level , 32(32.0%) persons that are
not record their sugar level and 16(16.0%) persons that response is
don’t know means that have not in mind that record their sugar level.
Among 52 persons that record their sugar level, the count (percentages)
for male and female were 19(36.5%) and 33(63.5%) respectively and among
32 persons that are not record their sugar level, the count
(percentages) for males and females were 16(50.0%) and 16(50.0%)
respectively and among 16 persons that don’t know means that have not in
mind that record their sugar level, the count (percentages) for male
and female were 4(25.0%) and 12(75.0%) respectively. Figure 3 shows that
of out of 1000 respondents, 43(43.0%) persons mostly their sugar level
remain normal , 39(39.0%) persons that are not their sugar level remain
normal and 18(18.0%) persons that response is don’t know means that have
not in mind that their sugar level remain normal. Among 43 persons that
their sugar level remain normal, the count (percentages) for male and
female were 18(41.9%) and 25(58.1%) respectively and among 39 persons
that are not their sugar level remain normal, the count (percentages)
for males and females were 14(35.9%) and 25(64.1%) respectively and
among 18 persons that don’t know means that have not in mind that their
sugar level remain normal, the count (percentages) for male and female
were 7(38.9%) and 11(61.1%) respectively. Figure 3 shows that of out of
1000 respondents, 28(28.0%) persons never fluctuate their sugar level,
45(45.0%) persons sometimes fluctuate their sugar level and 27(27.0%)
persons every time fluctuate their sugar level Among 28 persons never
fluctuate their sugar level, the count (percentages) for male and female
were 16(57.1%) and 12(42.9%) respectively and among 45 persons
sometimes fluctuate their sugar level, the count (percentages) for males
and females were 18(40.0%) and 27(60.0%) respectively and among 27
persons that every time fluctuate their sugar level, the count
(percentages) for male and female were 5(18.5%) and 22(81.5%)
respectively.
Figure 3 shows that of out of 1000 respondents, 52(52.0%) persons
that agree that diabetes hindrance in daily activities, 31(31.0%)
persons that not agree that diabetes hindrance in daily activities and
17(17.0%) persons that response answer in don’t know means they have no
idea that diabetes hindrance in daily activities. Among 52 that agree
that diabetes hindrance in daily activities, the count (percentages) for
male and female were 20(38.5%) and 32(61.5%) respectively and among 31
persons that not agree that diabetes hindrance in daily activities, the
count (percentages) for males and females were 11(35.5%) and 20(64.5%)
respectively and among 17 persons that response answer in don’t know
means they have no idea that diabetes hindrance in daily activities, the
count (percentages) for male and female were 8(47.1%) and 9(53.9%)
respectively. Figure 3 shows that of out of 1000 respondents, 50(50.0%)
persons that agree that health interfere hobbies or recreational
activities, 32(32.0%) persons that not agree that health interfere
hobbies or recreational activities and 18(18.0%) persons that response
answer in don’t know means they have no idea that health interfere
hobbies or recreational activities. Among 50 that agree that health
interfere hobbies or recreational activities, the count (percentages)
for male and female were 16(32.0%) and 34(68.0%) respectively and among
32 persons that not agree that health interfere hobbies or recreational
activities, the count (percentages) for males and females were 16(50.0%)
and 16(50.0%) respectively and among 18 persons that response answer in
don’t know means they have no idea that health interfere hobbies or
recreational activities, the count (percentages) for male and female
were 7(38.9%) and 11(61.1%) respectively. Figure 3 shows that of out of
1000 respondents, 47(47.0%) persons that agree that diabetes affected
daily life, 35(35.0%) persons that not agree that diabetes affected
daily life and 18(18.0%) persons that response answer in don’t know
means they have no idea that diabetes affected daily life. Among 47 that
agree that diabetes affected daily life, the count (percentages) for
male and female were 15(31.9%) and 32(68.1%) respectively and among 35
persons that not agree that diabetes affected daily life, the count
(percentages) for males and females were 17(48.6%) and 18(51.4%)
respectively and among 18 persons that response answer in don’t know
means they have no idea that health diabetes affected daily life, the
count (percentages) for male and female were 7(38.9%) and 11(61.1%)
respectively. Figure 3 shows that of out of 1000 respondents, 55(55.0%)
persons that agree that health interfere in household chores, 32(32.0%)
persons that not agree that health interfere in household chores and
13(13.0%) persons that response answer in don’t know means they have no
idea that health interfere in household chores. Among 55 that agree that
health interfere in household chores, the count (percentages) for male
and female were 16(29.1%) and 39(70.9%) respectively and among 32
persons that not agree that health interfere in household chores, the
count (percentages) for males and females were 16(50.0%) and 16(50.0%)
respectively and among 13 persons that response answer in don’t know
means they have no idea that health interfere in household chores, the
count (percentages) for male and female were 7(53.8%) and 6(46.2%)
respective. Figure 3 shows that of out of 1000 respondents, 37(37.0%)
persons that agree that diabetes affected social life, 43(43.0%) persons
that not agree that diabetes affected social life and 20(20.0%) persons
that response answer in don’t know means they have no idea that
diabetes affected social life. Among 37 that agree that diabetes
affected social life, the count (percentages) for male and female were
6(16.2%) and 31(83.8%) respectively and among 43 persons that not agree
that diabetes affected social life, the count (percentages) for males
and females were 26(60.5%) and 17(39.5%) respectively and among 20
persons that response answer in don’t know means they have no idea that
health diabetes affected social life, the count (percentages) for male
and female were 7(35.0%) and 13(65.0%) respectively.
Figure 3 shows that of out of 1000 respondents, 0(0.0%) persons that
are taking alcohol daily and 2(2.0%) persons that are taking alcohol
weekly and 5(5.0%) persons that are taking alcohol monthly and 93(93.0%)
persons are taking no alcohol. Among 0 that are taking alcohol daily,
the count (percentages) for male and female were 0(0.0%) and 0(0.0%)
respectively and among 2 persons that are taking alcohol weekly, the
count (percentages) for males and females were 2(100%) and 0(0.0%)
respectively and among 5 persons that are taking alcohol monthly, the
count (percentages) for male and female were 4(80.0) and 1(20.0) and 93
persons that are not taking alcohol, the count (percentages) for male
and female were 33(35.5) and 60(64.5) respectively. Figure 3 shows that
of out of 1000 respondents, 75(75.0%) persons that are living in non-
industrial area and 5(5.0%) persons that are living near the tyre
industry and 3(3.0%) persons that are living near the textile industry
and 5(5.0%) persons that are living near the steel mill and 12(12.0%)
persons are living near the other industries. Among 75 that are living
in non- industrial area, the count (percentages) for male and female
were 29(38.7%) and 46(61.3%) respectively and among 5 persons that are
living near the tyre industry, the count (percentages) for males and
females were 3(60.0%) and 2(40.0%) respectively and among 3 persons that
are living near the textile industry, the count (percentages) for male
and female were 2(66.7%) and 1(33.3%) and 5 persons that are living near
the steel mill, the count (percentages) for male and female were
1(20.0) and 4(80.0%) respectively and among 12 persons are living near
the other industries, the count (percentages) for male and female were
4(33.3) and 8(66.7%) respectively. Figure 3 shows that of out of 1000
respondents, 27(27.0%) persons that their area sanitary system is very
good and 45(45.0%) persons that their area sanitary system is good and
17(17.0%) persons that there area sanitary system is bad and 11(11.0%)
persons that their area sanitary system is very bad. Among persons that
their area sanitary system is very good, the count (percentages) for
male and female were 4(14.8%) and 23(85.2%) respectively and among 45
persons that their area sanitary system is good, the count (percentages)
for males and females were 27(60.0%) and 18(40.0%) respectively and
among persons that there area sanitary system is bad, the count
(percentages) for male and female were 7(41.2%) and 10(58.8%) and 11
persons that their area sanitary system is very bad, the count
(percentages) for male and female were 1(9.1) and 10(90.9%)
respectively. Figure 3 shows that of out of 1000 respondents, 60(60.0%)
persons that taking pills in medicine, 20(20.0%) persons that are taking
insulin in medicine and 20(20.0%) persons that taking combination of
pills and insulin in medicine. Among 60 persons that taking pills in
medicine, the count (percentages) for male and female were 24(41.4%) and
34(58.6%) respectively and among 20 persons that are taking insulin in
medicine, the count (percentages) for males and females were 24(40.0%)
and 36(60.0%) respectively and among 20 persons that taking combination
of pills and insulin in medicine, the count (percentages) for male and
female were 5(25.0%) and 15(75.0%) respectively.
Figure 3 shows that of out of 1000 respondents, 77(77.0%) persons
that taking medicine regularly, 17(17.0%) persons that are not taking
medicine and 6(6.0%) persons that miss sometime medicine. Among 77
persons that taking medicine regularly, the count (percentages) for male
and female were 25(32.5%) and 52(67.5%) respectively and among 17 %)
persons that are not taking medicine, the count (percentages) for males
and females were 10(58.8%) and 7(41.2%) respectively and among 6 persons
that miss sometime medicine, the count (percentages) for male and
female were 4(66.7%) and 2(33.3%) respectively. Figure 3 shows that of
out of 1000 respondents, 56(56.0%) persons that use of vitamins or
supplements, 43(43.0%) persons that are not use of vitamins or
supplements and 1(1.0%) persons that sometime use of vitamins or
supplements. Among 56 that use of vitamins or supplements, the count
(percentages) for male and female were 20(35.7%) and 36(64.3%)
respectively and 43 persons that are not use of vitamins or supplements,
the count (percentages) for males and females were 18(41.9%) and
25(58.1%) respectively among 1 persons that sometime use of vitamins or
supplements, the count (percentages) for males and females were
1(100.0%) and 0(0.0%) respectively . Figure 3 shows that of out of 1000
respondents, 5(5.0%) persons that meet their doctor weekly, 70(70.0%)
persons that meet their doctor monthly and 25(25.0%) persons that meet
their doctor yearly. Among 5 that persons that meet their doctor weekly,
the count (percentages) for male and female were 2(40.0%) and 3(60.0%)
respectively and 70 persons that meet their doctor monthly, the count
(percentages) for males and females were 28(40.0%) and 42(60.0%)
respectively among persons that meet their doctor yearly, the count
(percentages) for males and females were 9(36.0%) and 16(64.0%)
respectively . Figure 3 shows that of out of 1000 respondents, 86(86.0%)
persons that discuss problem in detail with doctor, 6(6.0%) persons
that are not discuss problem in detail with doctor and 8(8.0%) persons
that answer is don’t know means they don’t want to share that discuss in
detail with doctor or not. Among 86 persons that discuss problem in
detail with doctor, the count (percentages) for male and female were
34(39.5%) and 52(60.5%) respectively and among 6%) persons that are not
discuss problem in detail with doctor, the count (percentages) for males
and females were 2(33.3%) and 4(66.7%) respectively and among 8 persons
that answer is don’t know means they don’t want to share that discuss
in detail with doctor or not, the count (percentages) for male and
female were 3(37.5%) and 5(62.5%) respectively.
Figure 3 shows that of out of 1000 respondents, 78(78.0%) persons
that satisfied with their treatment, 12(12.0%) persons that are not
satisfied with their treatment and 10(10.0%) persons that response is
don’t know means they don’t want to share that are satisfied or not.
Among 78 persons that satisfied with their treatment, the count
(percentages) for male and female were 28(35.9%) and 50(64.1%)
respectively and among 12 persons that are not satisfied with their
treatment, the count (percentages) for males and females were 6(50.0%)
and 6(50.0%) respectively and among 10 persons that response is don’t
know means they don’t want to share that are satisfied or not, the count
(percentages) for male and female were 5(50.0%) and 5(50.0%)
respectively
Descriptive Analysis
In this section the frequency and percentages of the
demographic, different variable of diabetes will be discussed with
respect to diabetes gender. We will discuss here the frequency and
percentages of demographic variables There are 1000 subjects. The
debate of the results will base on the frequency, percentages.
Discussion
There were 39 males and 61 female’s people in sample of 1000.
Percentage of male persons=39.0%, Percentage of female persons=61.0%.
Out of 1000 respondents the number(percentage) of marital status in
single and married group was 25(25.0%) and 75(75.0%) respectively. Out
of 1000 respondents the number(percentage) of family members in 1-5,
6-10, 11-15 and 16-20 group was 37(37.0%),47(47.0%),12(12.0%) and
4(4.0%). Out of 1000 respondents the number(percentage) of other
diabetic patient in family in 1-2, 3-4, 5-6 and No group was 34(34.0%),
8(8.0%), 3(3.0%) and 55(55.0%) respectively. Out of 1000 respondents the
number(percentage) of Persons address in towns, local areas and out of
Lahore group was 54(54.0%), 35(35.0%) and 11(11.0%) respectively. Out of
1000 respondents the number(percentage) of persons that following any
exercise in yes and no group was 80(80.0%) and 20(20.0%) respectively.
Out of 1000 respondents the number(percentage) of Persons that have skin
problem after diabetes in yes, no and don’t know group was 17(17.0%),
79(79.0%) and 4(4.0%) respectively. Out of 1000 respondents the
number(percentage) of Persons that have wound healing problem after
diabetes in yes, no and don’t know group was 36(36.0%), 56(56.0%) and
8(8.0%) respectively. Out of 1000 respondents the number(percentage) of
Persons that have kidney problem after diabetes in yes, no and don’t
know group was 18(18.0%), 74(74.0%) and 8(8.0%) respectively. Out of
1000 respondents the number(percentage) of Persons that have vision
problem after diabetes in yes, no and don’t know group was 66(66.0%),
29(29.0%) and 5(5.0%) respectively. Out of 1000 respondents the
number(percentage) of Persons have weight loss after diabetes in yes, no
and don’t know group was 47(47.0%), 39(39.0%) and 14(14.0%)
respectively. Out of 1000 respondents the number(percentage) of Persons
have weight gain after diabetes in yes, no and don’t know group was
20(20.0%), 66(66.0%) and 16(16.0%) respectively. Out of 1000 respondents
the number(percentage) of Persons that hoteling in yes and no group was
37(37.0%), 63(63.0%) respectively.
Out of 1000 respondents the number(percentage) of Persons that taking
kind of meal in Wheat, Rice and Fiber group was 84(84.0%), 13(13.0%)
and 3(3.0%) respectively. Out of 1000 respondents the number(percentage)
of Persons that number of taken meal in a day in 1, 2, 3 and 4 group
were 1(1.0%) 19(19.0%), 71(71.0%) and 9(9.0%) respectively. Out of 1000
respondents the number(percentage) of Persons that are frequently
hoteling in Never, Sometimes, Normally and Frequently group was
64(64.0%), 23(23.0%) ,8(8.0%) and 5(5.0%) respectively. Out of 1000
respondents the number(percentage) of Persons that regularly test their
blood sugar level in yes, no and don’t know group was 51(51.0%),
42(42.0%) and 7(7.0%) respectively. Out of 1000 respondents the
number(percentage) of Persons that check their sugar in a day in once a
day, twice a day, Weekly and Monthly group was 8(8.08%), 20(20.0%),
46(46.0) and 26(26.0%) respectively. Out of 1000 respondents the
number(percentage) of Persons that fluctuate their sugar in never,
sometimes and every time group was 28(28.0%), 45(45.0%), 27(27.0)
respectively. Out of 1000 respondents the number(percentage) of Persons
living in industrial area Yes and NO group was 25(25.0%) and 75(75.0%)
respectively. Out of 100 respondents the number(percentage) of Persons
living near the which factory in None, Tyre industry, Textile industry,
Steel and Others group was 75(75.0%), 5(5.0%), 3(3.0%), 5(5.0%),
12(12.0%) respectively .Out of 1000 respondents the number(percentage)
of Persons living area in rural area and urban area group was 23(23.0%)
and 77(77.0%) respectively.
Out of 1000 respondents the number(percentage) of Persons satisfied
their sanitary system in very good, good, bad and very bad group was
27(27.0%), 45(45.0%), 17(17.0%) and 11(11.0%) respectively .Out of 1000
respondents the number(percentage) of Persons that use which type of
water in tap and filter group was 42(42.0%), 58(58.0%) respectively. Out
of 1000 respondents the number(percentage) of Persons that taking kind
of medicine pills, insulin and combination group was 60(60.0%),
20(20.0%) and 20(20.0%) respectively. Out of 1000 respondents the
number(percentage) of Persons that regularly take medicine in yes, no
and miss sometimes group was 77(77.0%), 17(17.0%) and 6(6.0%)
respectively. Out of 1000 respondents the number(percentage) of Persons
that are used vitamin or supplements in yes, no and sometime group was
56(56.0%), 43(43.0%) and 1(1.0%) respectively. Out of 1000 respondents
the number(percentage) of Persons that meet their doctor in Weekly,
Monthly, and yearly group was 5(5.0%), 70(70.0%) and 25(25.0%)
respectively. Out of 1000 respondents the number(percentage) of Persons
take alcohol in yes and no group was 6(6.0%) and 94(94.0%). Out of 1000
respondents the number(percentage) of Persons smoking in yes and no
group was 22(22.0%) and 78(78.0%). Out of 1000 respondents the
number(percentage) of Persons that are going for daily walk in yes, no
and do not know group was 79(79.0%), 21(21.0%) respectively.
Out of 1000 respondents the number(percentage) of Persons that think
the exercise is necessary for diabetic patients in yes, no and do not
know group was 80(80.0%), 17(17.0%) and 3(3.0%) respectively. Out of
1000 respondents the number(percentage) of Persons that think routine
walk is helpful for diabetic patients in yes, no and do not know group
was 88(88.0%), 9(9.0%) and 3(3.0%) respectively. Out of 1000 respondents
the number(percentage) of Persons follow doctor regarding exercise in
yes, no and don’t know group was 67(67.0%), 32(32.0%) and 1(1.0%)
respectively Out of 1000 respondents the number(percentage) of Persons
that take proper fruit in yes, no and sometime group was 41(41.0%),
20(20.0%) and 39(39.0%) respectively. Out of 1000 respondents the
number(percentage) of Persons that take milk regularly in yes, no, and
sometime group was 48(48.0%), 43(43.0%) and 34(34.0%) respectively. Out
of 1000 respondents the number(percentage) of Persons that skip their
meal in yes, no and sometime group was 37(37.0%), 34(34.0%) and
29(29.0%) respectively. Out of 1000 respondents the number(percentage)
of Persons that take care of their diet in yes, no and don’t know group
was 71(71.0%), 20(20.0%) and 9(9.0%) respectively. Out of 1000
respondents the number(percentage) of Persons that affected their daily
life from diabetes in yes, no and don’t know group was 47(47.0%),
35(35.0%) and 18(18.0%) respectively. Out of 1000 respondents the
number(percentage) of Persons that their household chores affected form
health in yes, no and don’t know group was 55(55.0%), 32(32.0%) and
13(13.0%) respectively. Out of 1000 respondents the number(percentage)
of Persons that spend the day for exercise in morning, afternoon,
evening and no group was 42(42.0%), 4(4.0%).31(31.0) and 23(23.0%)
respectively. Out of 1000 respondents the number(percentage) of Persons
that walking time in morning, afternoon, evening and no group was
52(52.0%), 4(4.0%). 23(23.0) and 21(21.0%) respectively. Out of 1000
respondents the number(percentage) of Persons that record their sugar
levels in yes, no and do not know group was 52(52.0%), 32(32.0%) and
16(16.0%) respectively. Out of 1000 respondents the number(percentage)
of persons that their sugar remains normal in yes, no and don’t know
group was 43(43.0%), 39(39.0%) and 18(18.0%) respectively. Out of 1000
respondents the number(percentage) of Persons that think diabetes become
hindrance in their daily walk activities in yes, no and do not know
group was 52(52.0%), 31(31.0%) and 17(17.0%) respectively. Out of 1000
respondents the number(percentage) of Persons that their health
interferes in their hobbies and recreational activities in yes, no and
don’t know group was 52(52.0%), 32(32.0%) and 18(18.0%) respectively.
Out of 1000 respondents the number(percentage) of Persons that their
social life affected from diabetes in yes, no and don’t know group was
47(47.0%), 35(35.0%) and 18(18.0%) respectively. Out of 1000 respondents
the number(percentage) of Persons that are frequently use alcohol in
daily, weekly, monthly and none group was 0(0.0%), 2(2.0%),5(5.0%) and
93(93.0%) respectively. Out of 100 0respondents the number(percentage)
of Persons that discuss their problems in detail with the doctor in yes,
no and don’t know group was 86(86.0%), 6(6.0%) and 8(8.0%)
respectively.
Read More About Lupine Publishers Journal of Current Trends on Biostatistics & Biometrics Please Click on the Below Link:
https://lupine-publishers-biostatistics.blogspot.com/