Lupine Publishers | Journal of Otolaryngology Research Impact Factor
Opinion
Project Planning Phase
b) Definition of research question(s).
c) Definition of data selection criteria, sampling variables and observation time(s) schedule.
d) Aspects of data collection and documentation, ongoing project quality controls.
e) Feasibility considerations of various project designs.
f) Administrative aspects: financials, selection of partners, estimation of required realization times.
g) Statistical methods for data analysis, feasibility of pilot- (sub)-projects.
h) Final definition of data selection criteria.
i) Aspects of results publication.
j) Aspects of possibly necessary actions in case of emergencies.
Project Realization Phase
b) Ongoing communication between all project partners.
c) Maintenance of open minds for early signs of project’s critical developments.
d) Ongoing monitoring for possible emergency actions.
e) The KISS principle: Keep everything as simple and stupid as possible.
I’d like to recommend using the items in the above project management as guidance for your project but under no condition as a comprehensive cookbook! As ENT scientist you should permanently remember that you are doing research in humans and not in bolts and nuts!
How you could easily detect the difference between a “user of statistical software” or a “real” statistician
The next table provides you with selected questions/topics to distinguish between professional statistician and software user (Table 1).
Table 1.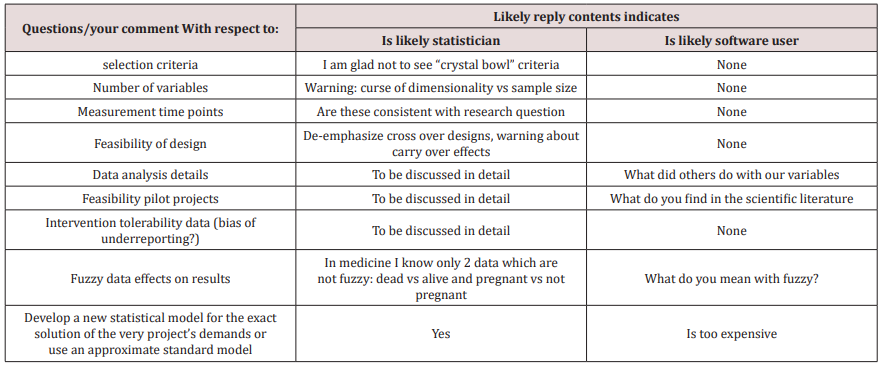
Read More Lupine Publishers Otolaryngology Journal Articles :
https://lupine-publishers-otolaryngology.blogspot.com/
Read More Lupine Publishers blogger Articles : https://lupinepublishers.blogspot.com

Read More Lupine Publishers Otolaryngology Journal Articles :
https://lupine-publishers-otolaryngology.blogspot.com/
Read More Lupine Publishers blogger Articles : https://lupinepublishers.blogspot.com

No comments:
Post a Comment
Note: only a member of this blog may post a comment.