Lupine Publishers| Journal of Material Science
Abstract
Brass is an important metalloid which is used in construction of sculptures. It is noticed that sculpture of brass is corroding due to interaction of pollutants. The pollutants develop chemical and electrochemical reaction on the surface of base material. Their concentrations of corrosive pollutants are increased in winter season. The air quality becomes very poor in winter season. Inside sculpture different forms of corrosion are observed like galvanic, pitting, stress, crevice etc. The major components of pollutants are oxides of carbon, oxides of nitrogen, oxides of sulphur, ammonia, ozone and particulates. Among these pollutants oxides of sulphur and ammonia are major corroder of brass. Ammonia is observed moist air to form ammonium hydroxide. It produces chemical reaction with brass metal and form complex compounds like [Zn(NH4)4](OH)2, [Zn(NH4)4]SO4, [Zn(NH4)]CO3, [Cu(NH4)4](OH)2, [Cu(NH4)4]SO4, [Cu(NH4)]CO3 etc. Oxides of sulphur react with moist air to exhibit sulphurous and sulphuric acids. They interact with brass to develop corrosion cell zinc metal and it is oxidized into Zn2+ ions and these ions are active to humidity and carbon dioxide to yield Zn(OH)2.ZnCO3.2H2O. Copper is converted into Cu2+ and it reacts with moist air and carbon dioxide to produce Cu(OH)2.Cu(CO3)2 and these complex compound detached on the surface of brass metal by rain water. These pollutants change their physical, chemical and mechanical properties and they also tarnish their facial appearance. Brass’ sculpture is affected by uniform corrosion. This type of corrosion can be control by nanocoating and electrospray techniques. For this work (6Z)-5,8-dihydrazono- 5,8-dibenzo[a,c][8]annulene and TiO2 are used as nanocoating and electrospray materials. The corrosion rate of material was determined by gravimetric and potentiostat technique. The nanocoating and electrospray compounds are formed a composite layer on surface of base metal. The formation of composite layer is analyzed by thermal parameters like activation energy, heat of adsorption, free energy, enthalpy and entropy. These thermal parameters were calculated by Arrhenius, Langmuir isotherm and transition state equations. Thermal parameters results are depicted that both materials are adhered with sculpture through chemical bonding. The surface coverage area and coating efficiency indicates that nanocoating and electrospray are produced a protective barrier in ammonia and sulphur dioxide atmosphere.
Keywords: Brass sculpture; Corrosion; Atmospheric pollutants; Nanocoating; Electrospray; Sulphur dioxide; Composite barrier
Introduction
The sculpture of brass comes in contact of contaminated air thus its deterioration starts for protection various types methods can be applied [1]. Brass [2] has major components is copper and zinc. Zn reacts the hot air to produce ZnO which is active in humidity [3] to convert into Zn(OH)2. In moist air [4], they form CuO, ZnO, Cu(OH)2 and Zn(OH)2. Both metals are active with sulphur to yield Cu2S, CuS and ZnS and these metallic sulphides [5] react with moist air to give Cu(OH)2, Zn(OH)2, CuSO4 and ZnSO4. The hydroxides of these metals interact with CO2 to produce CuCO3 and ZnCO3. Sulphur dioxide [6] is a culprit of brass. It undergoes with Cu(OH)2 and Zn(OH)2 to convert into CuSO4 and ZnSO4. Moist SO2 yields H2SO3 and H2SO4 whereas they create acidic environment [7] for brass and generate corrosion cell on their surface. It accelerates disintegration [8] in metal components of sculpture of brass. Brass is highly sensitive to ambient of ammonia gas [9]. It interacts with humid atmosphere [10] to NH4OH and it deposits on the surface brass metal [11] thus it converts into a complex layer of [Cu(NH3)4] (OH)2 and [Zn(NH3)4](OH)2 that layer erosion starts in rain water. [Cu(NH3)4](OH)2 and [Zn(NH3)4](OH)2 complex compounds [12] come in contact of H2SO4 environment to produce [Cu(NH3)4]SO4 and [Zn(NH3)4]SO4 that complex layer is eroded in rain water. In acidic medium brass outer face has developed CuSO4 and ZnSO4 when dust particulates [13] are deposited on their surface which contains Fe to remove Cu and Zn from outer surface. Dust particulates are possessed oxides of alkali metal in presence of moisture, it produces NaOH or KOH [14] that is create hostile environment for Zn and it forms complex compound [15] Na2[Zn(OH)4]or Na[Zn(OH)3.H2O] or Na[Zn(OH)3.(H2O)3]. The oxides of NO2 reacts with moist air to give HNO3 that acid produces chemical reaction with Cu and it converted into Cu(NO3)2. Some organic acids [16] available in air like acetic acid which develop corrosive environment for Cu and Zn which converts Cu into Cu2(CH3COO)4.H2O and Zn into (CH3COO)6. Zn4O complex compounds [17]. They are eroded by rain water on the surface of brass. Organic compounds [18] like amnio and sulpur increased day by day in atmosphere. They develop hostile environment for brass and corroding it. Corrosive pollutants [19] concentrations like oxides of carbon, oxides of nitrogen, oxides of sulphur, hydride of sulphur and nitrogen, ozone and particulates are enhanced due to industrials wastes, effluents, flues and other factors are like burning of coals, woods and cow dung cakes. Harmful pollutants [20] come into atmosphere through agricultural wastes, human wastes, pharmaceutical wastes, household wastes, food wastes and decomposition of living things. Various types of transports like road, water and air are evolving CO, NO2 and SO2 gases which produce acidic environments for brass. Several types of techniques are used to control the corrosion of brass like metallic coating; polymeric coating, paint coating, organic and inorganic coating of materials but these didn’t give satisfactory results in corrosive medium. Some organic and inorganic inhibitors are applied to protect the corrosion of materials in acidic but they provide good results. Hot dipping, electroplating and galvanization techniques is used as protective tools for brass corrosion in acidic medium but these methods don’t shave base metals. In this work it is to mitigate corrosion of brass corrosion by nanocoating and filler techniques. These materials form composite barrier on the surface base metal and blocked porosities and stop diffusion or osmosis process of pollutants.
Experimental
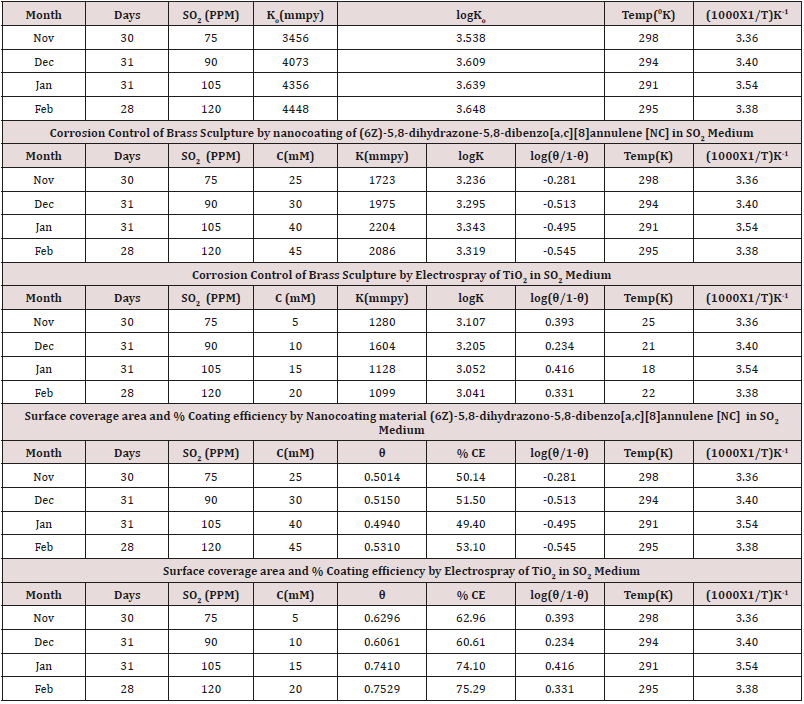
Brass coupons 15sqcm were taken for experimental analysis. Samples surface were rubbed with emery paper, rinsed with acetone, dry them and kept into desiccators. Sample kept 20meter height of roof in open sky and it observed that colour of brass can be changed. Corrosion rate was determined in winter season by weight loss method. The concentration of SO2 in November 75ppm, December 90ppm, January 105ppm and February 120ppm and temperatures recorded in this period were 298K, 294K, 291K and 295K. Synthesis organic compound (6Z)-5,8-dihydrazono- 5,8-dibenzo[a,c][8]annulene used as nanocoating and TiO2 as filler and corrosion of brass metal calculated in above mentioned concentrations and temperatures in winter season. Both compounds formed a composite barrier on surface of base metal (Figures 1-4). Surface adsorption phenomenon studied by thermal parameters like activation energy, heat of adsorption, free energy, enthalpy and entropy.Potentiostat/Galvanostat model EG&G used for corrosion potential, corrosion current and corrosion current density. Brass sample put between H2|Pt electrode as auxiliary electrode and Hg2Cl2|HgCl2 electrode reference electrode.
Synthesis of (6Z)-5,8-dihydrazono-5,8-dibenzo[a,c][8] annulene
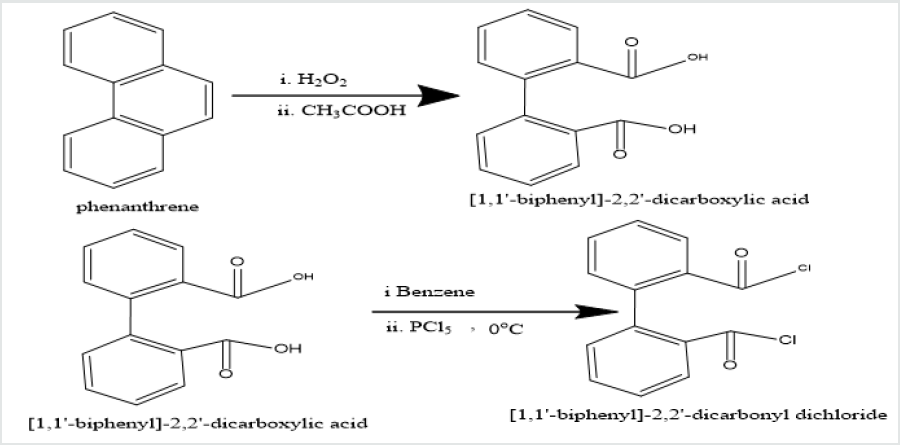
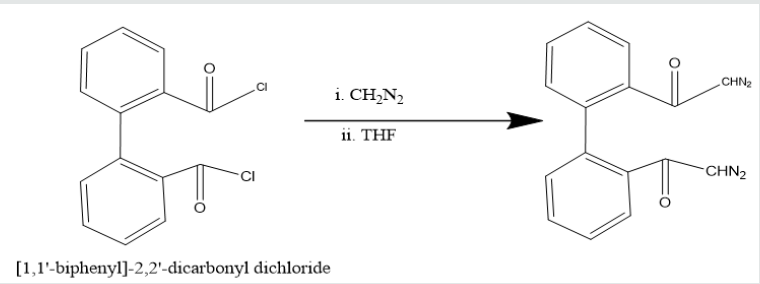
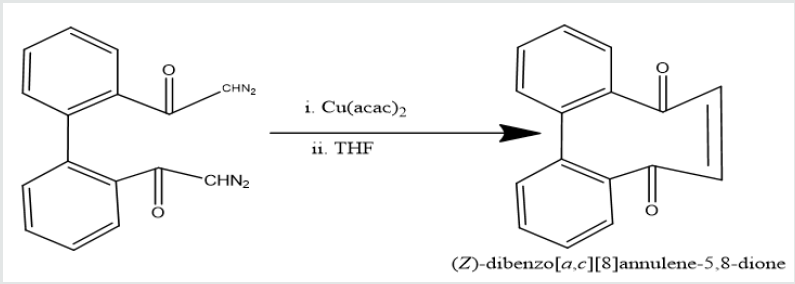
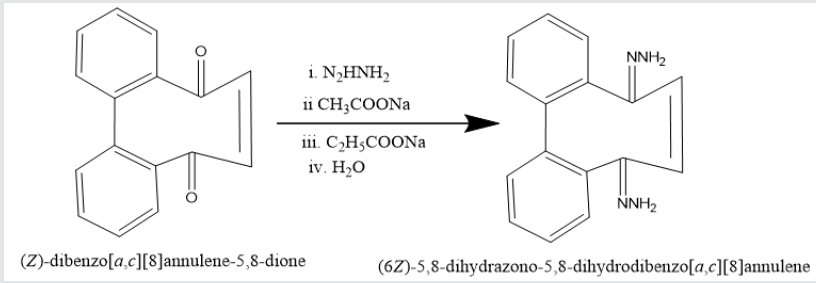
Phenatharene was oxidized into [1,1’-biphenyl]-2,2’- dicarboxylic acid by the use of H2O2 in presence of CH3COOH. When [1,1’-biphenyl]-2,2’-dicarboxylic acid was treated in PCl5 in benzene solution at 0 oC temperature, [1,1’-biphenyi]-2,2’-dicarbonyl chloride was obtained. It reacted with diazomethane to produce yield [1,1’-biphenyl]-2,2’-dicarboxodiazomethan which heated Cu(acac)2 in presence THF to yield (Z)-dibenzo[a,c][8]annulene- 5,8-dione. It was used with hydrazine hydrate in ethyl alcohol to give (6Z)-5,8-dihydrazone-5,8-dihydrodibenzo[a,c][8]annulene.
Results and Discussion
Brass metal was exposed in moist SO2 environment in 75ppm, 90ppm, 105ppm and 120ppm concentrations and 298 0K, 294 0K, 291 0K and 295 0K temperatures. The corrosion rate of brass metal was determined in winter season without coating and with coating (6Z)-5,8-dihydrazone-5,8-dihydrodibenzo[a,c][8]annulene and TiO2 electrospray of by weight loss formula K= 534 W/DAT (where W is weight loss, D is density and T is time) and their values were mentioned in (Table 1)
The corrosion rate of brass metal was recorded in the months of November, December, January and February, the results (Table 1) was shown that corrosion rate of metal increased in January to February but theses values were reduced with coating and filler materials like (6Z)-5,8-dihydrazone-5,8-dihydrodibenzo[a,c][8] annulene and TiO2. It was clearly noticed in (Figure 5) K versus Month. Brass metal kept into 75ppm, 90ppm, 105ppm and 120ppm of SO2 medium in month of Nov, Dec, Jan and Feb without coating. It was coated with 25mM, 30mM, 40mM and 45mM concentrations of (6Z)-5,8-dihydrazone-5,8-dibenzo[a,c][8]annulene, and again kept into same concentrations of SO2. After coating of (6Z)-5, 8-dihydrazone-5,8-dibenzo[a,c][8]annulene electrospray coating of TiO2 used at 5mM, 10mM, 15mm and 20mM concentrations and same concentrations SO2 Nov to Feb. The corrosion rates of in these three cases were written in (Table 1). These results were shown that corrosion rates without coating increased, it values decreased coating with (6Z)-5, 8-dihydrazone-5,8-dibenzo[a,c][8]annulene but their values more reduced with TiO2 electrospray. These trends were shown in (Figure 6) which plotted K versus C. The corrosion rates of brass metal at different temperatures 298 0K, 294 0K, 291 0K and 295 0K without and with coating were recorded in (Table 1). The addition of nanocoating and electrospray were reduced the corrosion rates as temperatures variation, it noticed in K versus T in (Figure 7).
Read More About Lupine Publishers Journal of Material Science Please Click on Below Link: https://lupine-publishers-material-science.blogspot.com/

No comments:
Post a Comment
Note: only a member of this blog may post a comment.